Kim Kardashian's Instagram Post Leads to FDA Warning

Kim Kardashian puts her baby bump on display while stopping by a studio on Tuesday afternoon (August 11) in Los Angeles.
The 34-year-old pregnant reality star’s sisters Kourtney and Khloe were also spotted at the studio that day.
It was reported that day that Kim‘s Instagram post endorsing a morning-sickness pill called Diclegis has led to the FDA sending the pharmaceutical company a warning.
The FDA says that the social media post, which has since been deleted, was “false and misleading” and “entirely omits all risk information.” Read more about the case on CNN.
Here is what Kim had written: “OMG. Have you heard about this? As you guys know my #morningsickness has been pretty bad … so I talked to my doctor. He prescribed me #Diclegis, and I felt a lot better and most importantly, it’s been studied and there was no increased risk to the baby. I’m so excited and happy with my results that I’m partnering with Duchesnay USA to raise awareness about treating morning sickness.”





































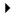
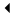 Older
Older





